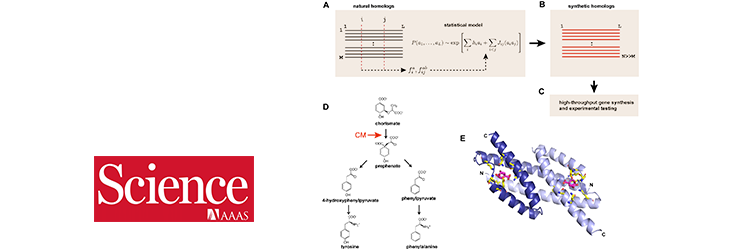
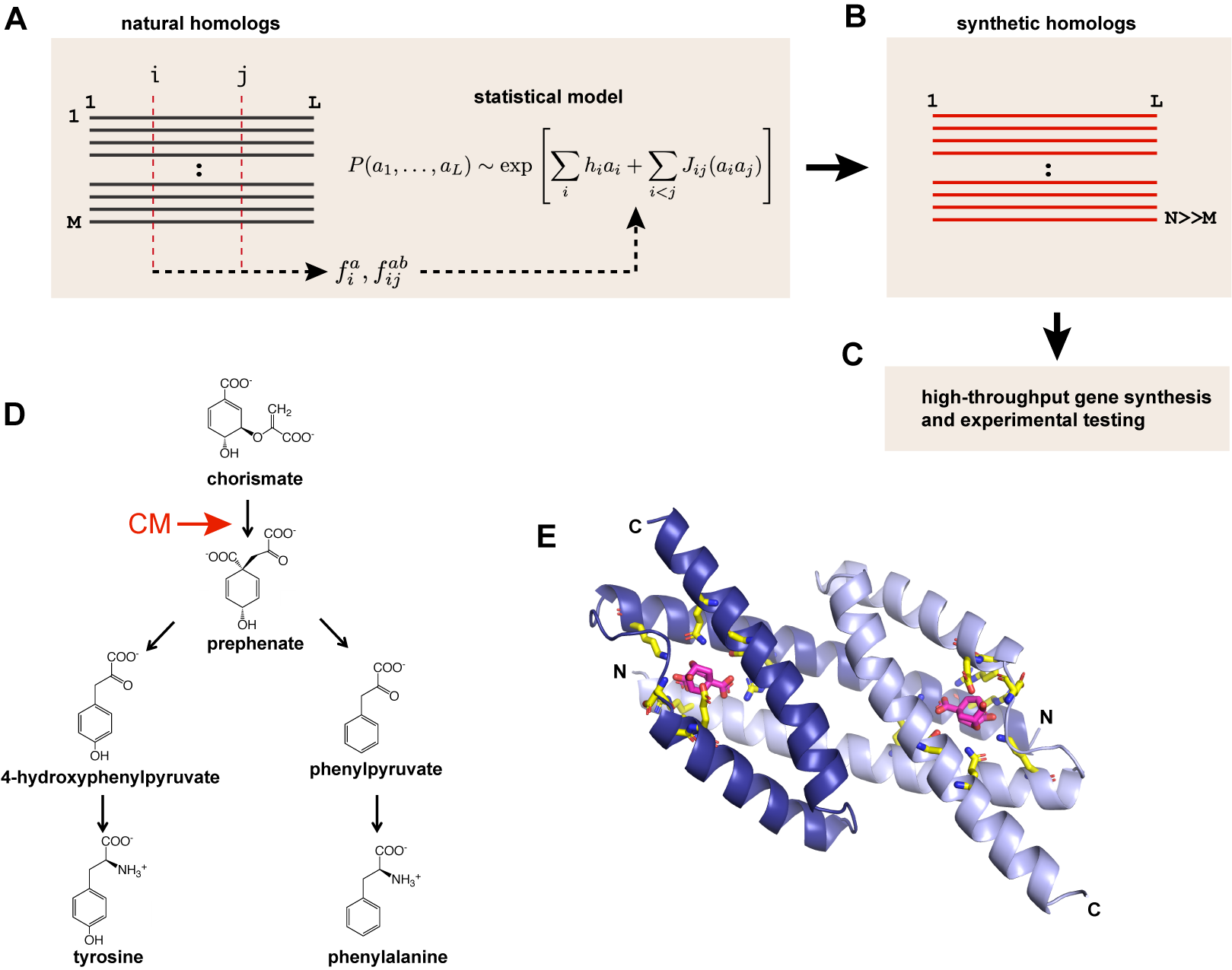
The rational design of enzymes is an important goal for both fundamental and practical reasons. Here, we describe a process to learn the constraints for specifying proteins purely from evolutionary sequence data, design and build libraries of synthetic genes, and test them for activity in vivo using a quantitative complementation assay. For chorismate mutase, a key enzyme in the biosynthesis of aromatic amino acids, we demonstrate the design of natural-like catalytic function with substantial sequence diversity. Further optimization focuses the generative model towards function in a specific genomic context. The data show that sequence-based statistical models suffice to specify proteins and provide access to an enormous space of functional sequences. This result provides a foundation for a general process for evolution-based design of artificial proteins.
Evolutionary data-driven protein engineering of Chorismate Mutase
More :
https://science.sciencemag.org/content/369/6502/440
Interview on ENS website “De la physique statistique pour modéliser des protéines”
Author affiliation :
Laboratoire de Physique de L’Ecole normale supérieure (LPENS, ENS Paris/CNRS/Sorbonne Université/Université de Paris)
Corresponding author : Rémi Monasson – Simona Cocco
Communication contact : L’équipe de communication